CBSE Class 11 Hydrogen Bonding Detail and Preparation Downloads
Hydrogen bonding, a subtle yet powerful force in the molecular realm, governs the behavior of countless substances. This unique intermolecular interaction arises when a hydrogen atom, covalently linked to a highly electronegative partner, forms an attractive bridge to another electronegative atom. Though individually weak, hydrogen bonds collectively shape the physical and chemical properties of materials, from water's anomalous characteristics to the stability of DNA's double helix. Unveiling the secrets of hydrogen bonding unveils a deeper understanding of nature's intricacies, influencing diverse fields such as biology, materials science, and pharmaceuticals. Join us in exploring the captivating world of molecular connections.
Exploring the Wonders of Hydrogen Bonding
What is hydrogen Bonding?
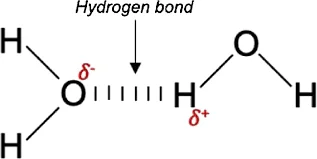
Hydrogen bonding is a special type of intermolecular force that occurs when a hydrogen atom, covalently bonded to a highly electronegative atom (such as oxygen, nitrogen, or fluorine), forms an attractive interaction with another electronegative atom in a different molecule. Despite being individually weak, hydrogen bonds collectively play a crucial role in shaping the properties of substances, influencing phenomena such as water's high boiling point, DNA's structure, and various biological and chemical processes. This unique bonding phenomenon contributes to molecules' cohesive and adhesive forces, impacting the physical and chemical behavior of a wide range of compounds.
Types of Hydrogen Bonding
Hydrogen bonding can be categorized into different types based on the molecular interactions. The primary types of hydrogen bonding include:
-
Hydrogen Bonding in Water (O-H...O):
- The most common type is seen in water molecules. Oxygen is more electronegative than hydrogen, creating a partial negative charge on oxygen and a partial positive charge on hydrogen. This leads to the formation of hydrogen bonds between neighboring water molecules.
-
Hydrogen Bonding in DNA (A-T, G-C):
- Within the DNA double helix, hydrogen bonding occurs between complementary base pairs—adenine (A) forms two hydrogen bonds with thymine (T), and guanine (G) forms three hydrogen bonds with cytosine (C).
-
Hydrogen Bonding in Proteins:
- Plays a crucial role in protein folding and stability. It involves hydrogen bonds between different amino acid residues within the protein structure.
-
Hydrogen Bonding in Amides and Amines:
- Amide groups (C=O) and amine groups (N-H) can form hydrogen bonds. This is often observed in compounds containing these functional groups.
-
Hydrogen Bonding in Carboxylic Acids:
- In carboxylic acids (RCOOH), hydrogen bonding occurs between the hydrogen of the hydroxyl group (–OH) and the oxygen of the carbonyl group (C=O).
-
Hydrogen Bonding in Alcohols:
- Alcohols (R-OH) can form hydrogen bonds, with the oxygen atom acting as the hydrogen bond acceptor and the hydrogen atom as the donor.
-
Hydrogen Bonding in Ammonia and Amines:
- Ammonia (NH₃) and amines (R₃N) can form hydrogen bonds with hydrogen bond acceptors such as water or other electronegative molecules.
Understanding these various types of hydrogen bonding is essential for comprehending the diverse roles this interaction plays in the properties and behaviors of different molecules across chemistry and biology.
Properties of Hydrogen Bonding
Hydrogen bonding imparts several unique and significant properties to substances, influencing their physical and chemical behavior. Here are some key properties associated with hydrogen bonding:
-
High Boiling and Melting Points:
- Hydrogen bonding leads to stronger intermolecular forces, requiring more energy to break these bonds. Substances with hydrogen bonding, such as water, typically have higher boiling and melting points.
-
High Surface Tension:
- Hydrogen bonding contributes to the cohesive forces between molecules, resulting in elevated surface tension in liquids like water. This is responsible for the formation of droplets and the capillary rise of liquids in narrow tubes.
-
High Heat of Vaporization:
- The energy required to convert a substance from a liquid to a gas (heat of vaporization) is higher in compounds with hydrogen bonding due to the need to break these strong intermolecular forces.
-
Anomalous Expansion of Water:
- Water exhibits an unusual behavior by expanding when it freezes. Hydrogen bonding causes water molecules to arrange in a hexagonal lattice structure, leading to a decrease in density and the floating of ice on liquid water.
-
Solubility of Polar Compounds:
- Hydrogen bonding enhances the solubility of polar compounds in water. Substances with hydrogen bond-forming groups can interact favorably with water molecules, leading to dissolution.
-
Stabilization of Biological Structures:
- Hydrogen bonds play a crucial role in the stability and structure of biological molecules. For example, they contribute to the double helix structure of DNA through base-pairing interactions.
-
Influence on Acidity and Basicity:
- Hydrogen bonding affects the acidity and basicity of compounds. Hydrogen ions involved in hydrogen bonding may exhibit different acidic or basic behavior compared to those not involved in such interactions.
-
Boiling Point Elevation in Solutions:
- When solutes are added to a solvent, hydrogen bonding in the solvent is disrupted, leading to an increase in boiling point. This phenomenon is commonly observed in solutions.
-
Facilitation of Chemical Reactions:
- Hydrogen bonding can influence the rate and outcome of chemical reactions, especially those involving reactive groups participating in hydrogen bond formation.
Understanding these properties of hydrogen bonding is crucial for appreciating its widespread influence in various chemical, physical, and biological processes.

CBSE Class 11th Downloadable Resources:
|
1. CBSE Class 11th Topic Wise Summary |
View Page / Download |
|
2. CBSE Class 11th NCERT Books |
View Page / Download |
|
3. CBSE Class 11th NCERT Solutions |
View Page / Download |
|
4. CBSE Class 11th Exemplar |
View Page / Download |
|
5. CBSE Class 11th Sample Papers |
View Page / Download |
|
6. CBSE Class 11th Question Bank |
View Page / Download |
|
7. CBSE Class 11th Topic Wise Revision Notes |
View Page / Download |
|
8. CBSE Class 11th Last Minutes Preparation Resources |
View Page / Download |
|
9. CBSE Class 11th Best Reference Books |
View Page / Download |
|
10. CBSE Class 11th Formula Booklet |
View Page / Download |
Being in CBSE class 11th and considering the board examinations you must be needing resources to excel in your examinations. At TestprepKart we take great pride in providing CBSE class 11th all study resources in downloadable form for you to keep you going.
Below is the list of all CBSE class 11th Downloads available on TestprepKart for both Indian and NRI students preparing for CBSE class 11th in UAE, Oman, Qatar, Kuwait & Bahrain.
SAMPLE PRACTICE QUESTIONS OF SIGNIFICANT FIGURES:
Q1. What is hydrogen bonding?
Answer. Hydrogen bonding is a special type of intermolecular force in which a hydrogen atom, covalently bonded to a highly electronegative atom, forms an attractive interaction with another electronegative atom in a different molecule. This creates a strong dipole-dipole interaction.
Q2. Why is water considered a unique substance due to hydrogen bonding?
Answer. Water exhibits a range of anomalous properties, such as high boiling and melting points, high surface tension, and expansion upon freezing, primarily due to the extensive hydrogen bonding between water molecules.
Q3. Can you provide examples of compounds exhibiting hydrogen bonding?
Answer. Yes, examples include water (H₂O), ammonia (NH₃), alcohols (e.g., ethanol), carboxylic acids (e.g., acetic acid), and biological molecules like DNA.
Q4. How does hydrogen bonding affect solubility?
Answer. Hydrogen bonding enhances the solubility of polar compounds in water. Molecules with hydrogen bond-forming groups can interact favorably with water molecules, promoting dissolution.
Q5. Why does boiling point increase in solutions with hydrogen bonding?
Answer. When solutes are added to a solvent, hydrogen bonding in the solvent is disrupted, leading to an increase in boiling point. This phenomenon is known as boiling point elevation.

| CBSE CLASS 11th Chemistry Chapters |
| Chapter1: SOME BASIC CONCEPTS OF CHEMISTRY |
| Chapter2: STRUCTURE OF ATOMS |
| Chapter3: CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES |
| Chapter4: CHEMICAL BONDING AND MOLECULAR STRUCTURE |
| > kossel lewis approach-to chemical bonding |
| > Ionic or Electrovalent Bond |
| > Bond Parameters |
| > The Valence Shell Electron Pair Repulsion (VSEPR) Theory |
| > Valence Bond Theory |
| > Hybridisation |
| > Molecular Orbital Theory |
| > Bonding in Some Homonuclear Diatomic Molecules |
| Chapter5: THERMODYNAMICS |
| Chapter6: EQUILIBRIUM |
| Chapter7: REDOX REACTIONS |
| Chapter8: ORGANIC CHEMISTRY - SOME BASIC PRINCIPLE AND TECHNIQUES |
| Chapter9: Hydrocarbons HYDROCARBONS |
| CBSE Class 11 Physics Chapters |
| Chapter1: UNITS AND MEASUREMENTS |
| Chapter2: MOTION IN A STRAIGHT LINE |
| Chapter3: MOTION IN A PLANE |
| Chapter4: LAWS OF MOTION |
| Chapter5: WORK, ENERGY AND POWER |
| Chapter6: SYSTEM OF PARTICLES AND ROTATIONAL MOTION |
| Chapter7: GRAVITATION |
| Chapter8: MECHANICAL PROPERTIES OF SOLIDS |
| Chapter9: MECHANICAL PROPERTIES OF FLUIDS |
| Chapter10: THERMAL PROPERTIES OF MATTER |
| Chapter12: KINETIC THEORY |
| Chapter13: OSCILLATIONS |
| Chapter14: WAVES |
| CBSE Class 11 Mathematics chapter |
| Chapter1: SETS |
| Chapter2: RELATIONS AND FUNCTIONS |
| Chapter3: TRIGONOMETRIC FUNCTIONS |
| Chapter4: COMPLEX NUMBER AND QUADRATIC EQUATIONS |
| Chapter5: LINEAR INEQUALITIES |
| Chapter6: PERMUTATIONS AND COMBINATIONS |
| Chapter7: BINOMIAL THEOREM |
| Chapter8: SEQUENCES AND SERIES |
| Chapter9: STRAIGHT LINES |
| Chapter10: CONIC SECTIONS |
| Chapter11: INTRODUCTION TO THREE-DIMENSIONAL GEOMETRY |
| Chapter12: LIMITS AND DERIVATIVES |
| Chapter13: STATISTICS |
| Chapter14: PROBABILITY |
| Class 8 Link soon |
| Class 9 Link soon |
| Class 10 Link soon |
| Class 12 Link soon |