CBSE Class 11 The Valence Shell Electron Pair Repulsion Theory Detail and Preparation Downloads
The Valence Shell Electron Pair Repulsion (VSEPR) Theory is a fundamental concept in chemistry that explains molecular geometry. Developed by Ronald Gillespie and Ronald Nyholm, it postulates that electron pairs around a central atom arrange themselves to minimize repulsion, determining the three-dimensional shape of a molecule. The theory relies on the idea that electron pairs, whether bonding or lone, repel each other, leading to specific geometric arrangements. By considering the number of electron pairs and bonding atoms, VSEPR provides a simple yet powerful tool for predicting molecular shapes, offering insights into the physical properties and behavior of diverse chemical compounds.
The Valence Shell Electron Pair Repulsion (VSEPR) Theory Unveiled
What Is VSEPR Theory?
The basic principle of VSEPR is that electron pairs, both bonding and non-bonding (lone pairs), around a central atom arrange themselves in space to minimize repulsion and achieve the most stable configuration. The repulsion arises from the negative charge carried by electrons, and the geometry of the molecule adjusts to keep these electron pairs as far apart as possible.
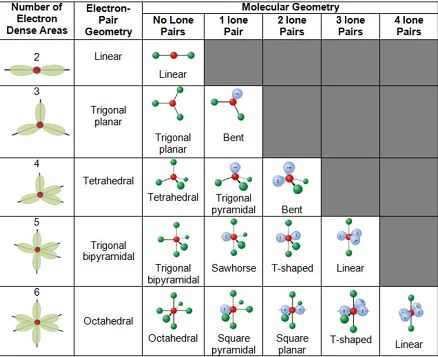
The key steps to applying VSEPR theory are:
-
Identify the central atom: Determine the atom in the molecule to which all other atoms are directly bonded.
-
Count electron pairs: Include both bonding pairs and lone pairs around the central atom.
-
Determine the molecular geometry: The electron pairs will arrange themselves to minimize repulsion. The possible geometries include linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral, among others.
-
Consider lone pairs: Lone pairs exert greater repulsion than bonding pairs, affecting the overall molecular shape.
-
Predict bond angles: Based on the geometry, predict the angles between the bonded atoms.
For example, in a molecule with a central atom surrounded by four electron pairs (tetrahedral geometry), if there are four bonding pairs, the molecule is methane (CH₄). If there are three bonding pairs and one lone pair, the molecule is ammonia (NH₃).
VSEPR theory is a valuable tool for understanding and predicting molecular shapes, which in turn influences the physical and chemical properties of substances. It is widely used in introductory chemistry and has practical applications in fields such as biochemistry and environmental science.
Postulates of VSEPR Theory
The Valence Shell Electron Pair Repulsion (VSEPR) theory is based on several postulates that help explain the geometry of molecules. These postulates are:
-
Electron Pair Repulsion: The primary postulate of VSEPR theory is that electron pairs, whether bonding or non-bonding (lone pairs), repel each other. This repulsion seeks to minimize the electron pair repulsion energy, leading to a specific arrangement of electron pairs around the central atom.
-
Geometry Determined by Electron Pair Arrangement: The three-dimensional geometry of a molecule is determined by the arrangement of electron pairs around the central atom. The goal is to position these pairs as far away from each other as possible to minimize repulsion.
-
Bonded and Lone Pairs are Treated Equally: In VSEPR theory, both bonded pairs and lone pairs of electrons are treated equivalently when determining the molecular geometry. Lone pairs, however, generally exert stronger repulsion than bonded pairs.
-
Repulsion Hierarchy: The repulsive forces between different types of electron pairs follow a hierarchy: lone pair-lone pair > lone pair-bond pair > bond pair-bond pair. This hierarchy influences the final molecular shape.
-
Predicting Molecular Shapes: By considering the number of bonding pairs and lone pairs around the central atom, one can predict the overall molecular shape or geometry. Common shapes include linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral.
-
Effective Pairs: The VSEPR theory introduces the concept of effective pairs, where multiple bonds (double or triple bonds) and single bonds are considered equivalent in determining the molecular geometry. For instance, a double bond is treated as a single effective pair of electrons.
-
Distorted Geometries: In certain cases, the presence of lone pairs can distort the idealized geometric shapes. This is particularly evident when lone pairs are present on the central atom, leading to deviations from the standard angles and shapes.
These postulates provide a foundation for understanding molecular shapes and predicting the arrangement of atoms in a molecule based on the repulsion between electron pairs. The VSEPR theory has proven to be a useful and widely applied tool in the field of chemistry.
Limitations of VSEPR Theory
While the Valence Shell Electron Pair Repulsion (VSEPR) theory is a valuable tool for predicting molecular geometries, it has certain limitations:
-
Lack of Consideration for Electron Pair Distortions: VSEPR assumes that electron pairs and atoms are spherical, which may not be entirely accurate. The theory doesn't consider the distortion of electron pairs due to the influence of nearby atoms, leading to deviations from predicted geometries.
-
Limited in Explaining Transition Metal Complexes: VSEPR is less effective in predicting the geometries of transition metal complexes where d-orbitals are involved. The theory doesn't account for the interactions between the metal center and the ligands in these cases.
-
Doesn't Address Molecular Orbitals: VSEPR doesn't consider molecular orbital theory, which provides a more accurate description of electron distribution in molecules. In cases where molecular orbital theory is more applicable, VSEPR may not offer a complete explanation.
-
Inability to Explain Some Observed Geometries: Some molecular geometries observed experimentally don't align with the predictions of VSEPR theory. For instance, certain molecules with expanded octets or unusual bond angles can't be accurately explained using VSEPR alone.
-
Disregard for Electron Pair Energy: VSEPR does not consider the energy associated with electron pairs. In reality, lone pairs may have a more significant impact on molecular stability and reactivity than VSEPR predicts.
-
Simplistic Treatment of Multiple Bonds: VSEPR treats multiple bonds as a single effective pair of electrons. While this simplification is useful, it doesn't fully capture the differences in repulsion between single and multiple bonds.
-
Applicability to Larger Molecules: VSEPR becomes increasingly challenging to apply accurately to larger and more complex molecules. The interaction of multiple atoms and electron pairs can lead to deviations from predicted geometries.
Despite these limitations, VSEPR remains a valuable and widely used model for introductory chemistry, providing a simple and intuitive way to predict molecular shapes. However, in more advanced and complex molecular systems, other theories and computational methods may be necessary for a more accurate description of molecular geometries and properties.

What Is the VSEP Number?
It seems there might be a small typo in your question. I assume you meant to ask about the VSEPR (Valence Shell Electron Pair Repulsion) number or VSEPR notation. If that's not what you were looking for, please provide more context or clarify your question.
| VSEP Number | Shape of the Molecule |
| 2 | Linear |
| 3 | Trigonal Planar |
| 4 | Tetrahedral |
| 5 | Trigonal Bipyramidal |
| 6 | Octahedral |
| 7 | Pentagonal Bipyramidal |
CBSE Class 11th Downloadable Resources:
|
1. CBSE Class 11th Topic Wise Summary |
View Page / Download |
|
2. CBSE Class 11th NCERT Books |
View Page / Download |
|
3. CBSE Class 11th NCERT Solutions |
View Page / Download |
|
4. CBSE Class 11th Exemplar |
View Page / Download |
|
5. CBSE Class 11th Sample Papers |
View Page / Download |
|
6. CBSE Class 11th Question Bank |
View Page / Download |
|
7. CBSE Class 11th Topic Wise Revision Notes |
View Page / Download |
|
8. CBSE Class 11th Last Minutes Preparation Resources |
View Page / Download |
|
9. CBSE Class 11th Best Reference Books |
View Page / Download |
|
10. CBSE Class 11th Formula Booklet |
View Page / Download |
Being in CBSE class 11th and considering the board examinations you must be needing resources to excel in your examinations. At TestprepKart we take great pride in providing CBSE class 11th all study resources in downloadable form for you to keep you going.
Below is the list of all CBSE class 11th Downloads available on TestprepKart for both Indian and NRI students preparing for CBSE class 11th in UAE, Oman, Qatar, Kuwait & Bahrain.
SAMPLE PRACTICE QUESTIONS OF SIGNIFICANT FIGURES:
Q1. What is the VSEPR theory?
Answer. The VSEPR theory is a model in chemistry that helps predict the three-dimensional geometry of molecules. It is based on the idea that electron pairs around a central atom arrange themselves to minimize repulsion, determining the overall shape of the molecule.
Q2. How does VSEPR theory work?
Answer. VSEPR theory works by considering the repulsion between electron pairs, both bonding and non-bonding, around a central atom. The theory aims to arrange these electron pairs in a way that minimizes repulsion, leading to specific geometric shapes.
Q3. Are there any limitations to VSEPR theory?
Answer. Yes, VSEPR theory has limitations, such as its simplistic treatment of electron pair repulsion, lack of consideration for electron pair distortions, and challenges in predicting molecular shapes in certain complex situations.
Q4. How does VSEPR theory explain molecular shapes with lone pairs?
Answer. Lone pairs of electrons exert stronger repulsion than bonding pairs. Their presence can influence the molecular geometry, causing deviations from the idealized shapes predicted by VSEPR theory.
Q5. Does VSEPR theory consider the actual sizes of atoms?
Answer. No, VSEPR theory simplifies the consideration of atomic sizes. It assumes that electron pairs and atoms are spherical and focuses on the repulsion between electron pairs
| CBSE CLASS 11th Chemistry Chapters |
| Chapter1: SOME BASIC CONCEPTS OF CHEMISTRY |
| Chapter2: STRUCTURE OF ATOMS |
| Chapter3: CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES |
| Chapter4: CHEMICAL BONDING AND MOLECULAR STRUCTURE |
| > kossel lewis approach-to chemical bonding |
| > Ionic or Electrovalent Bond |
| > Bond Parameters |
| > Valence Bond Theory |
| Chapter5: THERMODYNAMICS |
| Chapter6: EQUILIBRIUM |
| Chapter7: REDOX REACTIONS |
| Chapter8: ORGANIC CHEMISTRY - SOME BASIC PRINCIPLE AND TECHNIQUES |
| Chapter9: Hydrocarbons HYDROCARBONS |
| CBSE Class 11 Physics Chapters |
| Chapter1: UNITS AND MEASUREMENTS |
| Chapter2: MOTION IN A STRAIGHT LINE |
| Chapter3: MOTION IN A PLANE |
| Chapter4: LAWS OF MOTION |
| Chapter5: WORK, ENERGY AND POWER |
| Chapter6: SYSTEM OF PARTICLES AND ROTATIONAL MOTION |
| Chapter7: GRAVITATION |
| Chapter8: MECHANICAL PROPERTIES OF SOLIDS |
| Chapter9: MECHANICAL PROPERTIES OF FLUIDS |
| Chapter10: THERMAL PROPERTIES OF MATTER |
| Chapter12: KINETIC THEORY |
| Chapter13: OSCILLATIONS |
| Chapter14: WAVES |
| CBSE Class 11 Mathematics chapter |
| Chapter1: SETS |
| Chapter2: RELATIONS AND FUNCTIONS |
| Chapter3: TRIGONOMETRIC FUNCTIONS |
| Chapter4: COMPLEX NUMBER AND QUADRATIC EQUATIONS |
| Chapter5: LINEAR INEQUALITIES |
| Chapter6: PERMUTATIONS AND COMBINATIONS |
| Chapter7: BINOMIAL THEOREM |
| Chapter8: SEQUENCES AND SERIES |
| Chapter9: STRAIGHT LINES |
| Chapter10: CONIC SECTIONS |
| Chapter11: INTRODUCTION TO THREE-DIMENSIONAL GEOMETRY |
| Chapter12: LIMITS AND DERIVATIVES |
| Chapter13: STATISTICS |
| Chapter14: PROBABILITY |
| CBSE Class 8 Link soon |
| CBSE Class 9 Link soon |
| CBSE Class 10 Link soon |
| CBSE Class 12 Link soon |

%20Theory%20ch%204%20topic%204.webp)


