CBSE Class 11 Electronic Configurations and Types of Elements Detail and Preparation Downloads
Embark on a captivating exploration of the fundamental building blocks of the universe with a deep dive into electronic configurations and the diverse realm of elements. At the heart of chemistry lies the intricate dance of electrons, shaping the properties and behaviors of each element. Unravel the mysteries of s-, p-, d-, and f-blocks in the periodic table, as we navigate the unique signatures that dictate the essence of elements. Join us on a journey through the science of arrangement, where electrons script the tales of atomic structure and unveil the fascinating diversity that defines the very essence of matter.
A Journey through Electronic Configurations and the Diversity of Elements
Electronic configurations
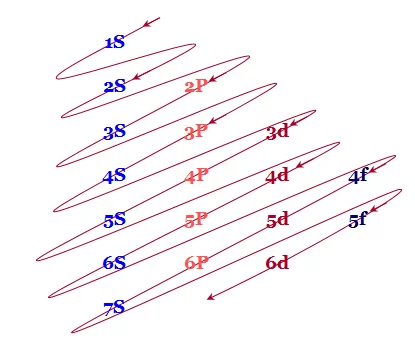
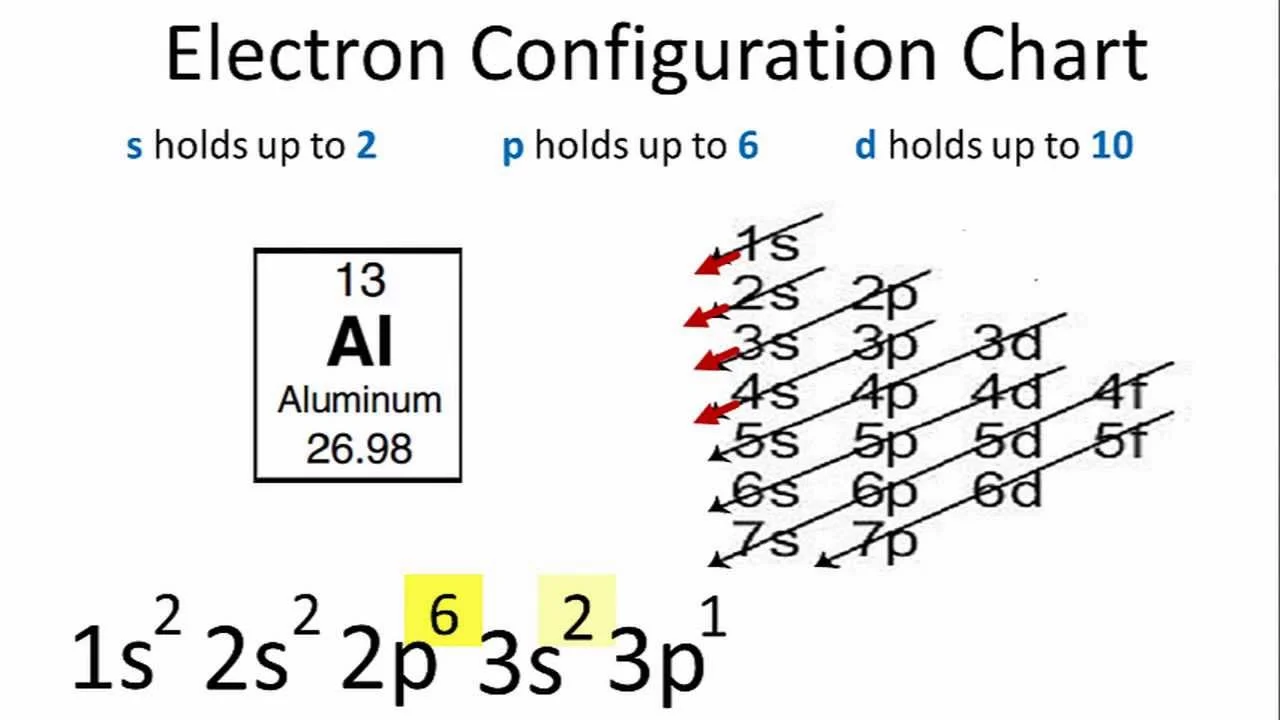
Electronic configurations are the quantum mechanical descriptions of the distribution of electrons within atoms. In simpler terms, they define how electrons are arranged in various energy levels (shells) and sublevels (orbitals) around an atomic nucleus. Each electron configuration is unique to an element and plays a crucial role in determining the element's chemical properties and behavior.
The notation follows a specific pattern, indicating the number of electrons in each sublevel. For instance, the electron configuration of carbon is 1s² 2s² 2p², denoting two electrons in the first energy level (1s²) and four in the second (2s² 2p²). Understanding electronic configurations is key to grasping the intricacies of chemical bonding and the periodic table.
What are Blocks?
In the periodic table, a "block" refers to a distinct region containing elements with similar electron configurations. These blocks are named after the type of atomic orbital in which the outermost electrons are found. The four main blocks are the s-block, p-block, d-block, and f-block. Each block exhibits specific trends and characteristics, influencing the behavior and properties of the elements within. From the highly reactive alkali metals in the s-block to the diverse properties of the transition metals in the d-block, understanding these blocks is fundamental to unraveling the intricacies of the periodic table and the behavior of its constituent elements
s-block
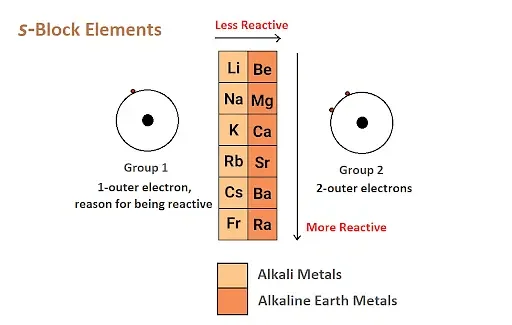
The s-block is a section of the periodic table containing elements in which the outermost electrons are placed in the s-orbital. This block consists of two groups: Group 1 (alkali metals) and Group 2 (alkaline earth metals). Elements in the s-block are known for their metallic properties, such as high electrical conductivity and luster.
Key characteristics of the s-block:
-
Location: The s-block is situated on the far left side of the periodic table.
-
Orbitals: Elements in the s-block fill their outermost s-orbital. For example, in the case of sodium (Na), its electron configuration is 1s² 2s² 2p⁶ 3s¹, indicating that the outermost electron is in the 3s orbital.
-
Reactivity: S-block elements are generally highly reactive, especially the alkali metals (Group 1), which are extremely reactive due to their tendency to lose their outermost electron easily.
-
Examples:
- Group 1: Hydrogen (H), Lithium (Li), Sodium (Na), Potassium (K), etc.
- Group 2: Beryllium (Be), Magnesium (Mg), Calcium (Ca), Strontium (Sr), Barium (Ba), etc.
-
Physical State: S-block elements exhibit a variety of physical states, ranging from gases (e.g., hydrogen, helium) to solids (e.g., sodium, potassium).
Understanding the properties of the s-block elements is crucial in comprehending the trends and behaviors exhibited by these elements in chemical reactions and their contributions to the overall periodic table.
p-block
The p-block is a region in the periodic table that includes elements whose outermost electrons are placed in the p-orbital. This block encompasses Groups 13 to 18 and consists of nonmetals, metalloids, and metals. Elements in the p-block exhibit diverse chemical properties and their electron configurations often end with p-orbital occupancies.
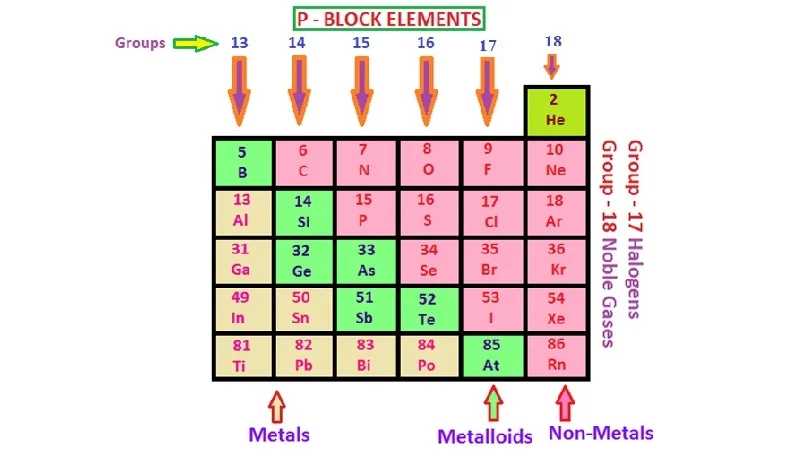
Key characteristics of the p-block:
-
Location: The p-block is found on the right side of the periodic table, after the s-block.
-
Orbitals: Elements in the p-block fill their outermost p-orbitals. For example, nitrogen (N) has the electron configuration 1s² 2s² 2p³, indicating that the outermost electrons are in the 2p orbitals.
-
Variety of Properties: The p-block includes elements with a wide range of properties, including nonmetals (e.g., nitrogen, oxygen), metalloids (e.g., boron, silicon), and metals (e.g., aluminum, sulfur).
-
Reactivity: P-block elements display diverse reactivity patterns. Nonmetals in this block often gain electrons, metalloids exhibit intermediate behavior, and metals tend to lose electrons.
-
Examples:
- Group 13: Boron (B), Aluminum (Al)
- Group 14: Carbon (C), Silicon (Si)
- Group 15: Nitrogen (N), Phosphorus (P)
- Group 16: Oxygen (O), Sulfur (S)
- Group 17: Fluorine (F), Chlorine (Cl)
- Group 18: Helium (He), Neon (Ne)
-
Physical States: P-block elements exist in various physical states, ranging from gases (e.g., nitrogen) to solids (e.g., sulfur) and include some important gases essential for life (e.g., oxygen).
Understanding the properties and trends of the p-block elements is crucial for predicting their behavior in chemical reactions and comprehending the overall diversity of the periodic table.
d-block
The d-block, also known as the transition metals, is a section of the periodic table that includes elements whose outermost electrons are placed in the d-orbital. This block spans from Group 3 to Group 12, fitting between the s-block and the p-block. Transition metals are characterized by their metallic properties, variable oxidation states, and the filling of the d-orbitals as electrons are added.

Key characteristics of the d-block:
-
Location: The d-block is located in the middle of the periodic table, between the s-block and the p-block.
-
Orbitals: Elements in the d-block fill their outermost d-orbitals. For example, iron (Fe) has the electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁶, indicating that the outermost electrons are in the 3d orbitals.
-
Variable Oxidation States: Transition metals are known for their ability to exhibit multiple oxidation states. The variable oxidation states arise from the filling of d-orbitals, allowing these elements to readily lose or gain electrons in chemical reactions.
-
Metallic Properties: D-block elements generally exhibit metallic properties such as high electrical conductivity, luster, and malleability.
-
Examples:
- Group 3: Scandium (Sc), Titanium (Ti)
- Group 4: Vanadium (V), Chromium (Cr)
- Group 5: Manganese (Mn), Iron (Fe)
- Group 6: Cobalt (Co), Nickel (Ni)
- Group 7: Copper (Cu), Zinc (Zn)
-
Catalytic Activity: Many transition metals in the d-block act as catalysts in chemical reactions due to their ability to adopt different oxidation states.
Understanding the properties of the d-block elements is essential for grasping their roles in various industrial processes, as well as their contributions to the magnetic and catalytic properties of materials.
f-block
The f-block is a region in the periodic table that includes two separate series of elements: the lanthanides and actinides. These elements are characterized by having their outermost electrons in the f-orbitals. The f-block is situated at the bottom of the periodic table and is often depicted as a detached row to conserve space.
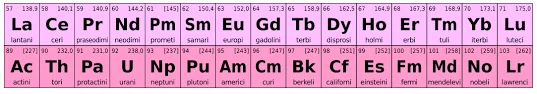
Key characteristics of the f-block:
-
Location: The f-block is located at the bottom of the periodic table, detached from the main body, and includes the lanthanides (from cerium to lutetium) and actinides (from thorium to lawrencium).
-
Orbitals: Elements in the f-block fill their outermost f-orbitals. For example, uranium (U) has the electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s² 4f¹⁴ 5d¹⁰ 6p⁶ 7s² 5f³ 6d¹, indicating that the outermost electrons are in the 5f orbitals.
-
Lanthanides and Actinides: The f-block is divided into the lanthanide series (from cerium to lutetium, including elements with atomic numbers 57 to 71) and the actinide series (from thorium to lawrencium, with atomic numbers 89 to 103).
-
Radioactive Properties: Many elements in the f-block, especially those in the actinide series, are radioactive. Uranium and plutonium are notable examples.
-
Applications: Actinides, such as uranium and plutonium, have practical applications in nuclear energy, while lanthanides find uses in technologies like catalysts, magnets, and phosphors.
-
Complex Chemistry: Elements in the f-block often exhibit complex and varied chemical behaviors, contributing to their importance in different fields of science and technology.
Understanding the f-block is essential for comprehending the unique properties and applications of these elements, particularly in the realms of nuclear science, materials science, and technology.

CBSE Class 11th Downloadable Resources:
|
1. CBSE Class 11th Topic Wise Summary |
View Page / Download |
|
2. CBSE Class 11th NCERT Books |
View Page / Download |
|
3. CBSE Class 11th NCERT Solutions |
View Page / Download |
|
4. CBSE Class 11th Exemplar |
View Page / Download |
|
5. CBSE Class 11th Sample Papers |
View Page / Download |
|
6. CBSE Class 11th Question Bank |
View Page / Download |
|
7. CBSE Class 11th Topic Wise Revision Notes |
View Page / Download |
|
8. CBSE Class 11th Last Minutes Preparation Resources |
View Page / Download |
|
9. CBSE Class 11th Best Reference Books |
View Page / Download |
|
10. CBSE Class 11th Formula Booklet |
View Page / Download |
Being in CBSE class 11th and considering the board examinations you must be needing resources to excel in your examinations. At TestprepKart we take great pride in providing CBSE class 11th all study resources in downloadable form for you to keep you going.
Below is the list of all CBSE class 11th Downloads available on TestprepKart for both Indian and NRI students preparing for CBSE class 11th in UAE, Oman, Qatar, Kuwait & Bahrain.
SAMPLE PRACTICE QUESTIONS OF SIGNIFICANT FIGURES:
Q1: What is an electronic configuration
Answer: Electronic configuration is the distribution of electrons in the atomic orbitals of an atom. It is represented as a series of numbers and letters that indicate the arrangement of electrons in different energy levels and sublevels.
Q2: How does the electronic configuration of an element relate to its position in the periodic table?
Answer: The electronic configuration of an element is closely tied to its position in the periodic table. Elements in the same group or column often share similar electronic configurations, influencing their chemical properties
Q3: What are the s-, p-, d-, and f-blocks in the periodic table?
Answer: These blocks refer to specific sections of the periodic table based on the type of atomic orbital in which the outermost electrons are located. The s-block includes Groups 1 and 2, the p-block spans Groups 13 to 18, the d-block consists of transition metals, and the f-block comprises the lanthanides and actinides.
Q4: How do electron configurations affect the properties of elements?
Answer: Electron configurations dictate an element's chemical behavior, reactivity, and physical properties. The arrangement of electrons in orbitals influences how elements interact in chemical reactions and form compounds.
Q5: How do electronic configurations help predict an element's behavior?
Answer: Electronic configurations provide insight into an element's valence electrons, allowing predictions about its reactivity, bonding tendencies, and chemical interactions. Similar electronic configurations often result in similar chemical properties.

| CBSE CLASS 11th |
| Chemistry Chapters |
| Chapter1: SOME BASIC CONCEPTS OF CHEMISTRY |
| Chapter2: STRUCTURE OF ATOMS |
| Chapter3: CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES |
| Why do we Need to Classify Elements? |
| > Genesis of Periodic Classification |
| > Modern Periodic Law and the Present Form of the Periodic Table |
| > Nomenclature of Elements with Atomic Numbers |
| > Electronic Configurations of Elements and the Periodic Table |
| > Periodic Trends in Properties of Elements |
| Chapter4: CHEMICAL BONDING AND MOLECULAR STRUCTURE |
| Chapter5: THERMODYNAMICS |
| Chapter6: EQUILIBRIUM |
| Chapter7: REDOX REACTIONS |
| Chapter8: ORGANIC CHEMISTRY - SOME BASIC PRINCIPLE AND TECHNIQUES |
| Chapter9: Hydrocarbons HYDROCARBONS |
| CBSE Class 11 Physics Chapters |
| Chapter1: UNITS AND MEASUREMENTS |
| Chapter2: MOTION IN A STRAIGHT LINE |
| Chapter3: MOTION IN A PLANE |
| Chapter4: LAWS OF MOTION |
| Chapter5: WORK, ENERGY AND POWER |
| Chapter6: SYSTEM OF PARTICLES AND ROTATIONAL MOTION |
| Chapter7: GRAVITATION |
| Chapter8: MECHANICAL PROPERTIES OF SOLIDS |
| Chapter9: MECHANICAL PROPERTIES OF FLUIDS |
| Chapter10: THERMAL PROPERTIES OF MATTER |
| Chapter12: KINETIC THEORY |
| Chapter13: OSCILLATIONS |
| Chapter14: WAVES |
| CBSE Class 11 Mathematics chapter |
| Chapter1: SETS |
| Chapter2: RELATIONS AND FUNCTIONS |
| Chapter3: TRIGONOMETRIC FUNCTIONS |
| Chapter4: COMPLEX NUMBER AND QUADRATIC EQUATIONS |
| Chapter5: LINEAR INEQUALITIES |
| Chapter6: PERMUTATIONS AND COMBINATIONS |
| Chapter7: BINOMIAL THEOREM |
| Chapter8: SEQUENCES AND SERIES |
| Chapter9: STRAIGHT LINES |
| Chapter10: CONIC SECTIONS |
| Chapter11: INTRODUCTION TO THREE-DIMENSIONAL GEOMETRY |
| Chapter12: LIMITS AND DERIVATIVES |
| Chapter13: STATISTICS |
| Chapter14: PROBABILITY |
| Class 8 Link soon |
| Class 9 Link soon |
| Class 10 Link soon |
| Class 12 Link soon |