CBSE Class 11 Electronic Configurations of Elements and the Periodic Table Detail and Preparation Downloads
his exploration delves into the captivating world of electronic configurations and the Periodic Table, where seemingly complex atomic structures are unveiled, and the periodicity of elements unfolds. Join us on this journey through the subatomic landscape, as we decipher the code that dictates the properties of elements, paving the way for a deeper understanding of the fundamental building blocks of matter. Welcome to the fascinating intersection of science and structure, where electrons choreograph the elegance of the periodic dance.
Electronic Configurations Unveiled Across the Periodic Table
What are Electron Configurations?
Electron configurations refer to the distribution of electrons among the various atomic orbitals in an atom. It describes the specific arrangement of electrons within the electron cloud surrounding the atomic nucleus. Understanding electron configurations is crucial in comprehending elements' behavior, chemical properties, and reactivity. The electron configuration of an atom is typically represented using a notation that indicates the principal energy level, the type of orbital, and the number of electrons in that orbital. This notation is based on the three quantum numbers that define an electron's position and energy within an atom.
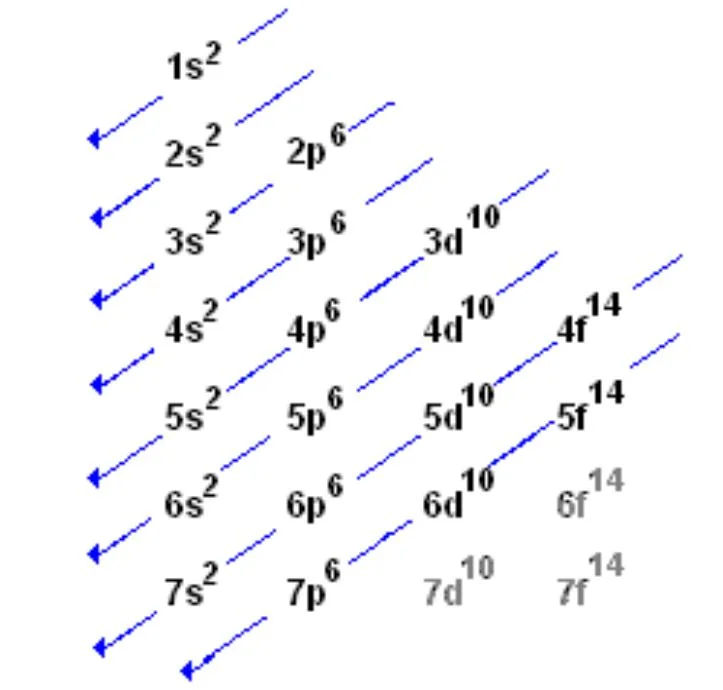
What is the Aufbau Principle?
The Aufbau Principle is a fundamental concept in quantum mechanics and chemistry that guides how electrons fill an atom's atomic orbitals. The word "Aufbau" is German for "building up," and the principle itself is often summarized as "filling up from the bottom."
The Aufbau Principle states:
Electrons occupy the lowest energy orbitals first before moving to higher energy orbitals.
In other words, when electrons are added to an atom, they enter the lowest energy orbitals available to them before occupying higher energy levels. The energy levels, or shells, are labeled with principal quantum numbers (n = 1, 2, 3, ...), and each level contains one or more sublevels or orbitals (s, p, d, f).
What is the Pauli Exclusion Principle?
The Pauli Exclusion Principle is a fundamental concept in quantum mechanics that was formulated by Austrian physicist Wolfgang Pauli in 1925. This principle addresses the behavior of identical particles with half-integer spins, such as electrons.
The Pauli Exclusion Principle states:
No two electrons (or other fermions with half-integer spins) in an atom can have the same set of four quantum numbers. As a result, no two electrons in an atom can occupy the same quantum state simultaneously. Furthermore, if two electrons occupy the same orbital, they must have opposite spins.
The four quantum numbers that describe an electron's state are:
- Principal Quantum Number (n): Represents the energy level of the electron.
- Angular Momentum Quantum Number (l): Describes the shape of the orbital.
- Magnetic Quantum Number (mₗ): Specifies the orientation of the orbital in space.
- Spin Quantum Number (mₛ): Describes the intrinsic angular momentum or spin of the electron.
Because electrons are fermions with a half-integer spin, the Pauli Exclusion Principle ensures that each orbital can accommodate a maximum of two electrons, and these electrons must have opposite spins.
What is Hund's rule?
Hund's Rule is a principle in quantum mechanics that guides the way electrons are distributed in atomic orbitals within an atom. This rule was formulated by German physicist Friedrich Hund and is one of the three fundamental principles, along with the Aufbau Principle and the Pauli Exclusion Principle, that govern the filling of electron orbitals.
Hund's Rule states:
Electrons occupy orbitals of the same energy level in a way that maximizes the total spin. Electrons will fill individual orbitals with parallel spins before pairing up.
In simpler terms:
- Electrons prefer to occupy separate orbitals within a subshell before pairing up.
- When electrons do pair up, they must have opposite spins.
The rule is based on the idea that electrons are negatively charged particles with intrinsic angular momentum, or spin.

What is the Shell?
In the context of atomic structure, a "shell" refers to a set of orbitals that have the same principal quantum number (n). The principal quantum number is one of the quantum numbers used to describe the energy levels of electrons in an atom. Each shell represents a different energy level, and electrons within a shell share similar energy.
What is the subshell?
Electrons within a subshell share the same values of the principal quantum number (n) and azimuthal quantum number (l). The azimuthal quantum number defines the shape of the orbital.
In a given shell (designated by the principal quantum number, n), there can be one or more subshells, each corresponding to a different value of the azimuthal quantum number (l). The values of l determine the specific type of orbital within a subshell:
- l = 0: The subshell is an s subshell, and it contains a single s orbital.
- l = 1: The subshell is a p subshell, and it contains three p orbitals.
- l = 2: The subshell is a d subshell, and it contains five d orbitals.
- l = 3: The subshell is an f subshell, and it contains seven f orbitals.
The number of orbitals in a subshell is equal to 2l + 1. For example, an s subshell (l = 0) has 2(0) + 1 = 1 orbital, a p subshell (l = 1) has 2(1) + 1 = 3 orbitals, and so on.
In summary, a subshell represents a collection of orbitals with similar shapes and energy levels within a particular electron shell.
| Principle Quantum Number Value | Value of Azimuthal Quantum Number | Resulting Subshell in the Electron Configuration |
| n=1 | l=0 | 1s |
|
n=2 |
l=0 | 2s |
| l=1 | 2p | |
|
n=3 |
l=0 | 3s |
| l=1 | 3p | |
| l=2 | 3d | |
|
n=4 |
l=0 | 4s |
| l=1 | 4p | |
| l=2 | 4d | |
| l=3 | 4f |
CBSE Class 11th Downloadable Resources:
|
1. CBSE Class 11th Topic Wise Summary |
View Page / Download |
|
2. CBSE Class 11th NCERT Books |
View Page / Download |
|
3. CBSE Class 11th NCERT Solutions |
View Page / Download |
|
4. CBSE Class 11th Exemplar |
View Page / Download |
|
5. CBSE Class 11th Sample Papers |
View Page / Download |
|
6. CBSE Class 11th Question Bank |
View Page / Download |
|
7. CBSE Class 11th Topic Wise Revision Notes |
View Page / Download |
|
8. CBSE Class 11th Last Minutes Preparation Resources |
View Page / Download |
|
9. CBSE Class 11th Best Reference Books |
View Page / Download |
|
10. CBSE Class 11th Formula Booklet |
View Page / Download |
Being in CBSE class 11th and considering the board examinations you must be needing resources to excel in your examinations. At TestprepKart we take great pride in providing CBSE class 11th all study resources in downloadable form for you to keep you going.
Below is the list of all CBSE class 11th Downloads available on TestprepKart for both Indian and NRI students preparing for CBSE class 11th in UAE, Oman, Qatar, Kuwait & Bahrain.
SAMPLE PRACTICE QUESTIONS OF SIGNIFICANT FIGURES:
Q1: What is electronic configuration?
Answer: Electronic configuration refers to the distribution of electrons among the various atomic orbitals within an atom. It provides a structured representation of how electrons occupy energy levels and subshells.
Q2: Why is electronic configuration important?
Answer: Electronic configuration is crucial in understanding an element's chemical properties, reactivity, and bonding behavior. It provides insights into the arrangement of electrons in an atom, influencing its overall behavior.
Q3: What is the Aufbau Principle?
Answer: The Aufbau Principle states that electrons fill the lowest energy orbitals first before moving to higher energy orbitals when filling an atom. It guides the order in which electrons occupy atomic orbitals.
Q4: What is Hund's Rule?
Answer: Hund's Rule states that electrons occupy orbitals of the same energy level singly before pairing up. This maximizes the total spin, contributing to the stability of the atom.
Q5: How does the Pauli Exclusion Principle apply to electronic configuration?
Answer: The Pauli Exclusion Principle states that no two electrons in an atom can have the same set of four quantum numbers. This principle enforces the rule that each orbital can accommodate a maximum of two electrons with opposite spins.

| CBSE CLASS 11th Chemistry Chapters |
| Chapter1: SOME BASIC CONCEPTS OF CHEMISTRY |
| Chapter2: STRUCTURE OF ATOMS |
| Chapter3: CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES |
| > Why do we Need to Classify Elements? |
| > Genesis of Periodic Classification |
| > Modern Periodic Law and the Present Form of the Periodic Table |
| > Nomenclature of Elements with Atomic Numbers |
| > Electronic Configurations and Types of Elements: 82 s-, p-, d-, f- Blocks |
| > Periodic Trends in Properties of Elements |
| Chapter4: CHEMICAL BONDING AND MOLECULAR STRUCTURE |
| Chapter5: THERMODYNAMICS |
| Chapter6: EQUILIBRIUM |
| Chapter7: REDOX REACTIONS |
| Chapter8: ORGANIC CHEMISTRY - SOME BASIC PRINCIPLE AND TECHNIQUES |
| Chapter9: Hydrocarbons HYDROCARBONS |
| CBSE Class 11 Physics Chapters |
| Chapter1: UNITS AND MEASUREMENTS |
| Chapter2: MOTION IN A STRAIGHT LINE |
| Chapter3: MOTION IN A PLANE |
| Chapter4: LAWS OF MOTION |
| Chapter5: WORK, ENERGY AND POWER |
| Chapter6: SYSTEM OF PARTICLES AND ROTATIONAL MOTION |
| Chapter7: GRAVITATION |
| Chapter8: MECHANICAL PROPERTIES OF SOLIDS |
| Chapter9: MECHANICAL PROPERTIES OF FLUIDS |
| Chapter10: THERMAL PROPERTIES OF MATTER |
| Chapter12: KINETIC THEORY |
| Chapter13: OSCILLATIONS |
| Chapter14: WAVES |
| CBSE Class 11 Mathematics chapter |
| Chapter1: SETS |
| Chapter2: RELATIONS AND FUNCTIONS |
| Chapter3: TRIGONOMETRIC FUNCTIONS |
| Chapter4: COMPLEX NUMBER AND QUADRATIC EQUATIONS |
| Chapter5: LINEAR INEQUALITIES |
| Chapter6: PERMUTATIONS AND COMBINATIONS |
| Chapter7: BINOMIAL THEOREM |
| Chapter8: SEQUENCES AND SERIES |
| Chapter9: STRAIGHT LINES |
| Chapter10: CONIC SECTIONS |
| Chapter11: INTRODUCTION TO THREE-DIMENSIONAL GEOMETRY |
| Chapter12: LIMITS AND DERIVATIVES |
| Chapter13: STATISTICS |
| Chapter14: PROBABILITY |
| Class 8 Link soon |
| Class 9 Link soon |
| Class 10 Link soon |
| Class 12 Link soon |

.webp)

