CBSE Class 11 Genesis of Periodic Classification Detail and Preparation Downloads
The inception of the periodic classification can be traced back to a time when the understanding of the fundamental building blocks of matter was in its infancy. The mid-19th century witnessed a surge of curiosity and experimentation as scientists grappled with the ever-growing list of known elements. The chemical landscape was becoming increasingly crowded, and the need for a systematic arrangement became paramount. One of the pioneers in this endeavor was Dmitri Mendeleev, a Russian chemist whose brilliance would leave an indelible mark on the scientific world.
The Genesis of Periodic Classification – A Comprehensive Guide for CBSE NCERT Download
Genesis of Periodic Classification
The genesis of periodic classification lies in the mid-19th century, marked by the collective efforts of brilliant minds like Mendeleev, Newlands, and Meyer. In 1869, Dmitri Mendeleev presented his revolutionary periodic table, organizing known elements based on atomic mass and predicting undiscovered ones. This pioneering work, guided by the Periodic Law, transformed the chaotic landscape of chemistry into a systematic and predictive framework. The collaborative spirit of these scientists laid the foundation for the elegant periodic classification we study today, shaping the course of modern chemistry.
Classification of Elements
The classification of elements is a fundamental concept in chemistry, organizing the diverse array of elements based on their properties. The periodic table, a visual representation of this classification, arranges elements in rows and columns according to atomic number, which reflects the number of protons in an atom's nucleus. This systematic arrangement allows scientists to discern patterns, trends, and relationships among elements, aiding in predictions of their behavior. Elements are grouped into periods and families, sharing similar characteristics. This classification is crucial for understanding the underlying principles of chemical reactions, atomic structure, and the diverse nature of the elements that constitute the building blocks of matter.
The Periodic Law
The Periodic Law is a fundamental principle in chemistry that states that the physical and chemical properties of elements are periodic functions of their atomic numbers. This law forms the basis of the periodic table, where elements are arranged in order of increasing atomic number, resulting in a systematic organization of elements with similar properties. Dmitri Mendeleev and Lothar Meyer independently formulated the Periodic Law in the late 19th century, leading to the development of the periodic table. This law enables scientists to predict and understand the behavior of elements based on their positions in the periodic table.
Elements
.webp)
Elements are the fundamental substances that constitute matter. Comprising atoms with a specific number of protons in their nuclei, each element possesses unique chemical properties. Represented by symbols on the periodic table, elements range from hydrogen to uranium. They combine to form compounds and molecules, contributing to the vast diversity of substances in the universe. Understanding elements is paramount in chemistry, serving as the foundation for comprehending the intricacies of reactions, structures, and the composition of the physical world.
What exactly is a Periodic Table?
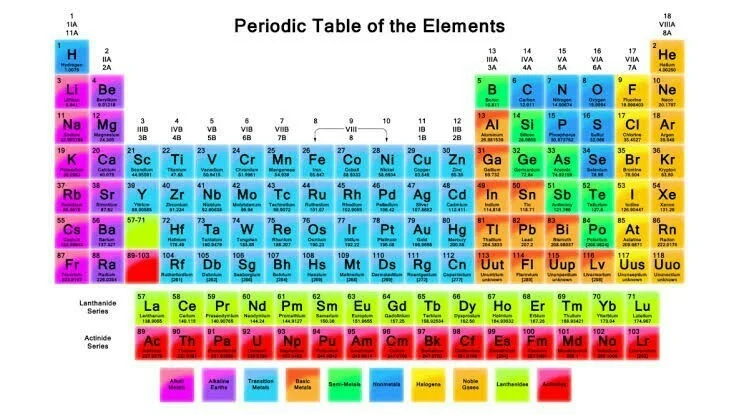
The Periodic Table is a tabular arrangement of chemical elements, organized based on their atomic number, electron configuration, and recurring chemical properties. Elements are presented in order of increasing atomic number, which corresponds to the number of protons in an atom's nucleus. The table is divided into periods (rows) and groups (columns). Each element is represented by its chemical symbol and elements within the same group share similar chemical properties. The Periodic Table is a powerful tool for understanding the relationships and patterns among elements, aiding in the prediction of their behavior and properties.
Newland’s law of Octaves
John Newlands proposed the Law of Octaves in the late 19th century as an early attempt to classify elements. According to this law, elements exhibit similar properties and characteristics when arranged in ascending order of atomic masses, with every eighth element repeating the properties of the first. He likened this pattern to the musical scale, where every eighth note sounds similar. While Newland's Law of Octaves was an early step in organizing elements, it had limitations and did not hold for all elements. Despite its shortcomings, it contributed to the eventual development of the more comprehensive periodic table.
Mendeleev Periodic Table
Dmitri Mendeleev's Periodic Table, unveiled in 1869, revolutionized the understanding of chemical elements. Organizing elements based on increasing atomic masses, Mendeleev arranged them into rows and columns, revealing a periodic pattern of properties. Notably, he left gaps for undiscovered elements, accurately predicting their properties. The periodic law, stating that properties recur periodically when elements are arranged by atomic mass, was a cornerstone of Mendeleev's table. His pioneering work laid the foundation for the modern periodic table, organized by atomic number, solidifying Mendeleev's legacy in the history of chemistry.
The Prout Hypothesis
The Prout Hypothesis, proposed by William Prout in the early 19th century, suggested that all elements are composed of hydrogen atoms, the simplest element. Prout speculated that other elements' atomic masses were multiples of hydrogen's mass. While the hypothesis failed due to inaccuracies in atomic mass measurements, it hinted at the concept of atomic weight and contributed to discussions on the nature of atomic structure. Ultimately, advancements in atomic theory, notably by John Dalton, disproved Prout's hypothesis, paving the way for a more nuanced understanding of atomic composition.

CBSE Class 11th Downloadable Resources:
|
1. CBSE Class 11th Topic Wise Summary |
View Page / Download |
|
2. CBSE Class 11th NCERT Books |
View Page / Download |
|
3. CBSE Class 11th NCERT Solutions |
View Page / Download |
|
4. CBSE Class 11th Exemplar |
View Page / Download |
|
5. CBSE Class 11th Sample Papers |
View Page / Download |
|
6. CBSE Class 11th Question Bank |
View Page / Download |
|
7. CBSE Class 11th Topic Wise Revision Notes |
View Page / Download |
|
8. CBSE Class 11th Last Minutes Preparation Resources |
View Page / Download |
|
9. CBSE Class 11th Best Reference Books |
View Page / Download |
|
10. CBSE Class 11th Formula Booklet |
View Page / Download |
Being in CBSE class 11th and considering the board examinations you must be needing resources to excel in your examinations. At TestprepKart we take great pride in providing CBSE class 11th all study resources in downloadable form for you to keep you going.
Below is the list of all CBSE class 11th Downloads available on TestprepKart for both Indian and NRI students preparing for CBSE class 11th in UAE, Oman, Qatar, Kuwait & Bahrain.
SAMPLE PRACTICE QUESTIONS OF SIGNIFICANT FIGURES:
Q1. What is the genesis of periodic classification?
Answer. The genesis of periodic classification refers to the historical development and evolution of systematic arrangements of chemical elements based on their properties, leading to the creation of the periodic table.
Q2. What is Mendeleev's contribution to the periodic table?
Answer. Dmitri Mendeleev's major contribution was the creation of a periodic table that arranged elements based on their atomic masses, predicting the properties of undiscovered elements and leaving gaps for them in his table.
Q3. How has the periodic table evolved?
Answer. The periodic table has evolved with advancements in atomic theory and experimental techniques. It continues to be refined and expanded as new elements are discovered and our understanding of atomic structure deepens.
Q4. What role did atomic number play in the evolution of the periodic table?
Answer. The discovery of atomic numbers, later attributed to Henry Moseley, led to the modern periodic table. Elements were reorganized based on atomic number rather than atomic mass, providing a more accurate reflection of the periodicity of properties.
Q5. How does the Periodic Law play a role in the genesis of periodic classification?
Answer. The Periodic Law, formulated by Mendeleev, states that the properties of elements are a periodic function of their atomic masses. This principle guided the systematic arrangement of elements in the periodic table.
| CBSE CLASS 11th Chemistry Chapters |
| Chapter1: SOME BASIC CONCEPTS OF CHEMISTRY |
| Chapter2: STRUCTURE OF ATOMS |
| Chapter3: CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES |
| > Why do we Need to Classify Elements? |
| > Modern Periodic Law and the Present Form of the Periodic Table |
| > Nomenclature of Elements with Atomic Numbers |
| > Electronic Configurations of Elements and the Periodic Table |
| > Electronic Configurations and Types of Elements: 82 s-, p-, d-, f- Blocks |
| > Periodic Trends in Properties of Elements |
| Chapter4: CHEMICAL BONDING AND MOLECULAR STRUCTURE |
| Chapter5: THERMODYNAMICS |
| Chapter6: EQUILIBRIUM |
| Chapter7: REDOX REACTIONS |
| Chapter8: ORGANIC CHEMISTRY - SOME BASIC PRINCIPLE AND TECHNIQUES |
| Chapter9: Hydrocarbons HYDROCARBONS |
| CBSE Class 11 Physics Chapters |
| Chapter1: UNITS AND MEASUREMENTS |
| Chapter2: MOTION IN A STRAIGHT LINE |
| Chapter3: MOTION IN A PLANE |
| Chapter4: LAWS OF MOTION |
| Chapter5: WORK, ENERGY AND POWER |
| Chapter6: SYSTEM OF PARTICLES AND ROTATIONAL MOTION |
| Chapter7: GRAVITATION |
| Chapter8: MECHANICAL PROPERTIES OF SOLIDS |
| Chapter9: MECHANICAL PROPERTIES OF FLUIDS |
| Chapter10: THERMAL PROPERTIES OF MATTER |
| Chapter12: KINETIC THEORY |
| Chapter13: OSCILLATIONS |
| Chapter14: WAVES |
| CBSE Class 11 Mathematics chapter |
| Chapter1: SETS |
| Chapter2: RELATIONS AND FUNCTIONS |
| Chapter3: TRIGONOMETRIC FUNCTIONS |
| Chapter4: COMPLEX NUMBER AND QUADRATIC EQUATIONS |
| Chapter5: LINEAR INEQUALITIES |
| Chapter6: PERMUTATIONS AND COMBINATIONS |
| Chapter7: BINOMIAL THEOREM |
| Chapter8: SEQUENCES AND SERIES |
| Chapter9: STRAIGHT LINES |
| Chapter10: CONIC SECTIONS |
| Chapter11: INTRODUCTION TO THREE-DIMENSIONAL GEOMETRY |
| Chapter12: LIMITS AND DERIVATIVES |
| Chapter13: STATISTICS |
| Chapter14: PROBABILITY |
| Class 8 Link soon |
| Class 9 Link soon |
| Class 10 Link soon |
| Class 12 Link soon |