I E Irodov Thermodynamics And Molecular Physics (The Second Law of Thermodynamics Entropy) Q . 2.152
I E Irodov Thermodynamics And Molecular Physics (The Second Law of Thermodynamics Entropy)
The Second Law of Thermodynamics Entropy is very important from the perspective to maximum score in IIT JEE. The Second Law of Thermodynamics expresses that the condition of entropy of the whole universe, as an isolated system, will consistently increment after some time.
The second law additionally expresses that the adjustments in the entropy in the universe can never be negative.
A good study paln is key for success, especially in JEE Mains and try to learn all formula. It should be your habit on daily basis. So, you can achieve good marks.
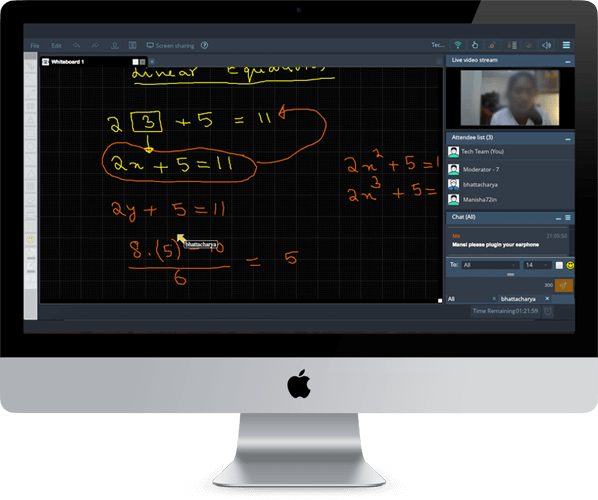
Q. 2.152 A piece of copper of mass m1 = 300g with initial temperature t1 = 97°C is placed into a calorimeter in which the water of mass m2=100 g is at a temperature t2=7 °C. Find the entropy increment of the system by the moment the temperatures equalize. The heat capacity of the calorimeter itself is negligibly small.
Complete I E Irodov Thermodynamics And Molecular Physics (The Second Law of Thermodynamics Entropy) Q. 2.152 Video Solution -
Indian & NRI students who wish to take admission in top engineering colleges in India, Knowing the important topics of the latest JEE Main, weightage & The Second Law of Thermodynamics Entropy video solutions will help Indian & NRI students prepare faster and score more.
If any of your questions are unanswered please ask your questions below in the comment box. We will give you the solutions to your questions.
Key Point Of The Second Law of Thermodynamics Entropy:
-
The second law of thermodynamics expresses that the all out entropy of a disconnected framework can never decrease over time, and is consistent if and just if all procedures are reversible. Isolated systems suddenly advance towards thermodynamic balance, the state with greatest entropy.
-
The total entropy of system and its environment can stay consistent in perfect situations where the framework is in thermodynamic equilibrium, or is experiencing an (imaginary) reversible procedure. In all procedures that happen, including spontaneous processes, the total entropy of the framework and its environmental factors increments and the procedure is irreversible in the thermodynamic sense.
-
The second law has been communicated from multiple points of view. Its first plan is credited to the French researcher Sadi Carnot, who in 1824 indicated that there is an upper limit to the efficiency of transformation of heat to work in a heat engine.
Important 'The Second Law of Thermodynamics Entropy' Questions You Should Not Miss:
|
I E Irodov Topics Of |
I E Irodov Solutions |
|
Thermodynamics and Molecular Physics The Second Law of Thermodynamics, Entropy Q. 2.117 |
|
|
Thermodynamics and Molecular Physics The Second Law of Thermodynamics, Entropy Q. 2.125 |
|
|
Thermodynamics and Molecular Physics The Second Law of Thermodynamics, Entropy Q. 2.14 |
|
|
Thermodynamics and Molecular Physics The Second Law of Thermodynamics, Entropy Q. 2.147 |
Important Topics Covered In The Second Law of Thermodynamics Entropy:
The second law was an empirical finding that was acknowledged as an axion of thermodynamic theory. statistical mechanics, traditional or quantum, clarifies the microscopic origin of the law.
Following topics are covered in I E Irodov solutions:
|
I E Irodov Topics |
I E Irodov Solutions |
|
Equation of the Gas State, Processes |
|
|
The first Law of Thermodynamics, Heat Capacity |
|
|
Kinetic Theory of Gases. Boltzmann's Law and Maxwell's |
|
|
Liquids. Capillary Effects |
|
|
Phase Transformations |
|
|
Transport Phenomena |
Important IE Irodov Video Solutions You Should Not Miss:
- Physical Fundamentals of Mechanics
- Electrodynamics
- Oscillations and Waves
- Optics
- Atomic and Nuclear Physics
Useful JEE Main Links You Should Not Miss:
JEE Details You Should Not Miss:
JEE Coaching Centers: